Background
Among the microorganisms scattering and floating in the air, there are viruses, bacteria, spore-forming bacteria, and fungi. These are known as the cause of infectious diseases even for the general healthy people and for the immune function decline person as well. On how to prevent this infection, what currently and widely utilized is the filtration using high-performance filter known as a HEPA filter (sterilizing). This has been introduced in hospitals as a device to prevent the infection by air cleaner or as an exhaust infection control unit.
In infection prevention measures by these HEPA filter, however there were some problems.
- Problems of infection prevention by HEPA filter
-
- The regular replacement of filter is required due to the clogging in the filter.
- The maintenance cost is very expensive for the filter replacement. Because bacteria and viruses have been collected in the filter and must be handled by the specialized supplier which is really costly.
- If proliferation filter replacement is delayed, there is a risk of breeding and proliferation of bacteria and viruses collected because they are not dead at all but still alive.
As a way to resolve these problems, we can learn from our ancestors that people killed microorganisms by overheating to prevent the spread of infection when the ancient epidemic occurred. Based on this fact,
the development of sterilization equipment
to kill microorganisms by heating process
started in 1998. After receiving a subsidy based on the SME business creation method, the heater development started. And in the following year 1999, we succeeded in developing 3D heater for the first time. Thereafter, we pursued the development of the heat exchanger for cooling off the exhaust air temperature. And finally we released the first product Perfect Clean in year 2003 and exhibited it at Lake Biwa Environmental Business Messe 2003. Having this reputation, we installed the first product to the St. Marianna University School of Medicine Hospital in the same year.
Then we concentrated on collecting the information to be reviewed as sterilization equipment and presentations at international exhibitions. In the year 2009, we together with Kyoto Prefectural University of Medicine applied the grant of Japan Science and Technology Agency for the immediate support to the local needs project. As it was granted, we succeeded in the spore-forming bacteria sterilization at 400°C or higher. Then we are funded by Kyoto SME Technology Development Promotion Project in 2011 and developed a new Perfect Clean adapting a collection of new technology such as the power system and the new air-cooled heat exchanger by flattening Palblat. At the same time, the academic "Incineration Sterilization Study Group" was established by the relevant academic and the industry leaders in order to clear the various technical issues towards the commercialization. And now we are fully promoting for its product commercialization.
Joint venture with Kyoto Prefectural University of Medicine
Demonstration of sterilization performance
Selection of bacteria to be used in the experiment
Selection criteria of microorganism:
- Size 5µm or less to cause the air infection
- Should survive in the dry state.
- Do not get sick even if they are infected.
- Easy to scatter.
As bacteria satisfying the above conditions
Bacillus subtilis natto (industrial natto bacteria spores)
Cell count of 109 per 1g (spore growth number)
were selected.

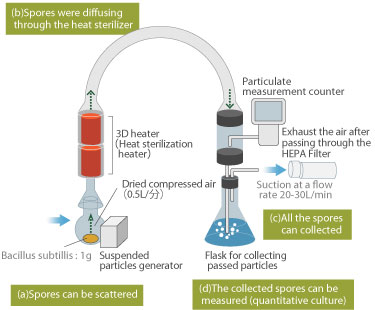
Configuration of the experimental model
The configuration of experimental model of incineration sterilization as follows:
- Apparatus to fly spores in the air
- Apparatus for burning the spores scattered (3D heater)
- Apparatus to recover the spores
- Culturing the spores recovered quantitatively and its measure
The experiment was executed with the above configuration
The sterilization experiment and the number of spore-forming bacteria
The sterilization equipment was operated under the following conditions:
- Heating range of 3D heater: 600°C or higher above the room temperature
- The duration of spores passing through the heat sterilization heater: less than 1 sec.
With the 3D heater at 7 different temperatures shown below, and the spore number of bacteria recovered are:
- Room temperature: 107
- 200°C: less than 107
- 250°C: less than 106
- 300°C: 102 ~ 103
- 350°C: without growth of the spore-forming bacteria
- 400°C: without growth of the spore-forming bacteria
- 600°C: without growth of the spore-forming bacteria
and as a result, the complete sterilization was confirmed at 350 °C
Prototype production toward the final product
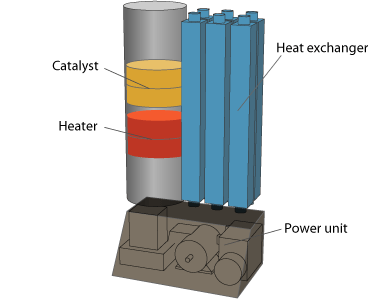
Heat exchange performance test of the prototype machine
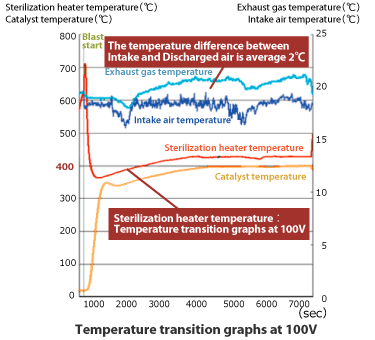
This is a prototype machine that combines the air-cooled heat exchanger system and the 3D heater. The temperature of the discharged air which was thermally incinerated and secured over 400°C for the sterilization was just plus 2°C over the air intake.